https://plantmolecularbiology.bio.lmu.de/index.html
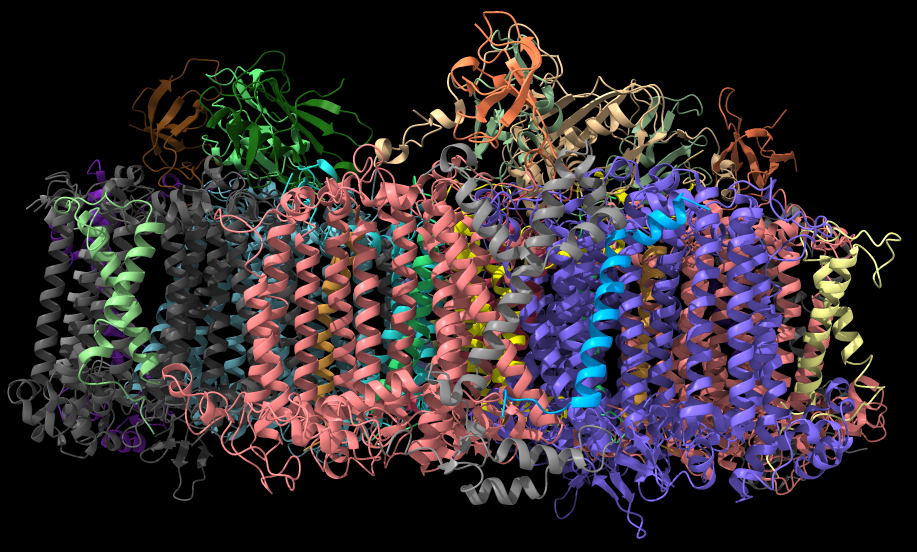
Oxygenic photosynthesis uses the energy of sunlight to generate the oxygen we breathe and the food we eat, but the vast majority of this energy is not converted to biomass. In fact, the light reactions of photosynthesis use only a fraction of the solar spectrum. Because they are recalcitrant to enhancement by conventional approaches, there is an urgent need to develop novel concepts to redesign their light harvesting and photochemical capacity. This includes the modification and exchange of entire ensembles of interacting photosynthesis components (see Figure 1) to design light reactions that can harvest and safely convert energy from an expanded solar spectrum.
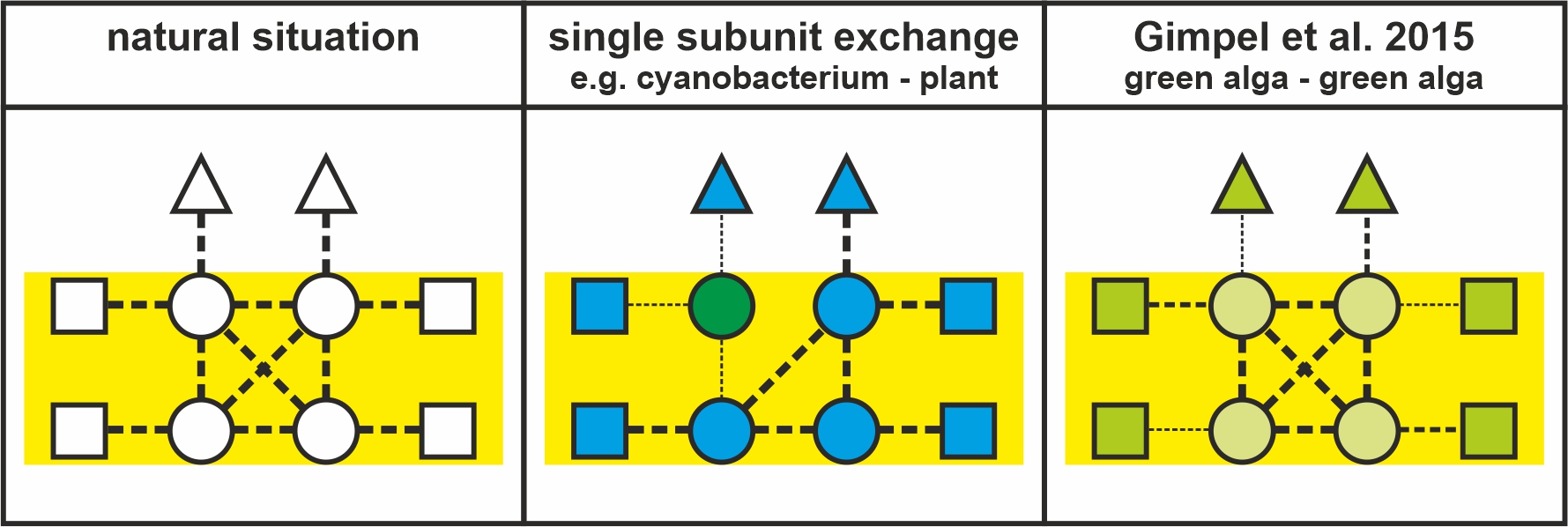
To this end synthetic biology and adaptive laboratory evolution are needed to combine photosystems and antenna systems from different photoautotrophic organisms. This may ultimately lead to including de novo-designed antennas in reimagined photosystems. As an adaptable chassis for this endeavour we use a cyanobacterial chassis.
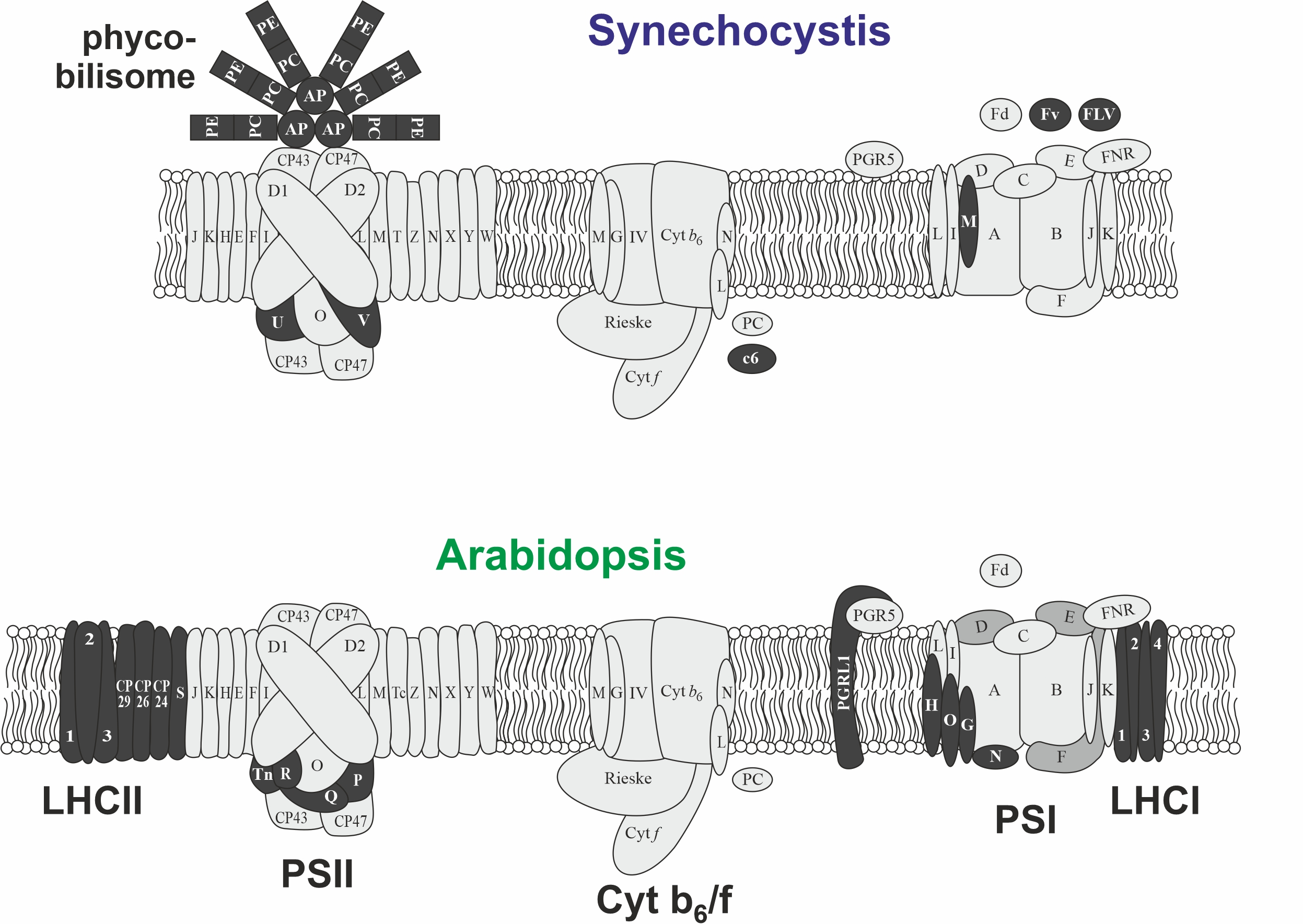
The cyanobacterium Synechocystis is the ideal test bed for enhancing the light reactions by synthetic biology and an experimentally-accessible proxy for eukaryotic photosynthesis hitherto considered to be immutable. This is possible because the basic structure of the photosynthetic machinery is conserved between cyanobacteria and plants, while peripheral components, in particular antennas have diversified (Figure 2). Moreover, Synechocystis is also accessible to adaptive laboratory evolution experiments (Dann et al. 2021) that should be required to enable strains provided with more light harvesting capacity to cope with the additional energy input. Finally, working on transferring entire multi-protein complexes by genetic engineering from one species to another species allows to tackle fundamental questions of how multi-protein complexes have evolved, how they do assemble and how assembly can occur before specific assembly factors have evolved.
References
- D. Leister, Enhancing the light reactions of photosynthesis: Strategies, controversies, and perspectives. Mol Plant. 2023 Jan 2;16(1):4-22
- M. Dann, E. M. Ortiz, M. Thomas, A. Guljamow, M. Lehmann, H. Schaefer, D. Leister, Enhancing photosynthesis at high light levels by adaptive laboratory evolution. Nat Plants 2021 , doi: 10.1038/s41477-021-00904-2
- M. Dann, D. Leister, Evidence that cyanobacterial Sll1217 functions analogously to PGRL1 in enhancing PGR5-dependent cyclic electron flow. Nat Commun 2019 , 10: 5299
- D. Leister, Thawing out frozen metabolic accidents. BMC Biol. 2019 , 17: 8.
- D. Leister, Genetic engineering, synthetic biology and the light reactions of photosynthesis. Plant Physiol 2019 ,179: 778-793
- M. Dann, D. Leister, Enhancing (crop) plant photosynthesis by introducing novel genetic diversity. Philos Trans R Soc Lond B Biol Sci. 2017 , 372: 20160380
- J. A. Gimpel, H. H. Nour-Eldin, M. A. Scranton, D. Li, S. P. Mayfield, Refactoring the six-gene photosystem II core in the chloroplast of the green algae Chlamydomonas reinhardtii. ACS Synth Biol. 2016 , 5: 589-96
- D. Leister, Experimental evolution in photoautotrophic microorganisms as a means of enhancing chloroplast functions. Essays Biochem 2018 , 62: 77-84

